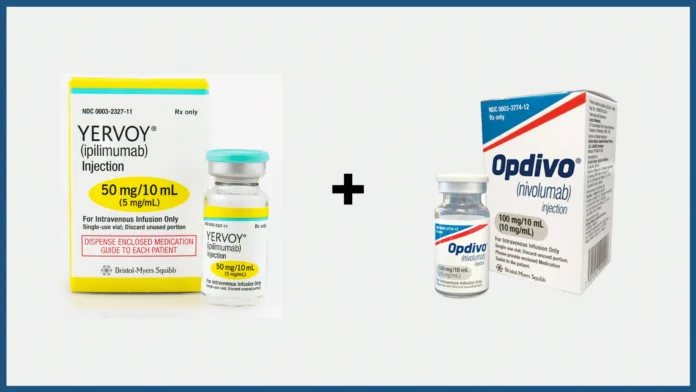
U.S. Food and Drug Administration Approves Opdivo® (nivolumab) plus Yervoy® (ipilimumab) as a First-Line Treatment for Unresectable or Metastatic Hepatocellular Carcinoma
Bristol Myers Squibb (NYSE: BMY) today announced that the U.S. Food and Drug Administration (FDA) approved Opdivo® (nivolumab) plus Yervoy® (ipilimumab) as a first-line treatment for adult patients with unresectable or metastatic hepatocellular carcinoma (HCC), the most common primary liver cancer.
This approval is based on the results from the global Phase 3 randomized, open-label CheckMate-9DW trial evaluating the combination of Opdivo plus Yervoy compared to investigator’s choice of tyrosine kinase inhibitor monotherapy (lenvatinib or sorafenib) in patients with unresectable or metastatic HCC who have not received prior systemic therapy.
In the trial, Opdivo plus Yervoy demonstrated statistically significant overall survival (OS) and overall response rate (ORR) vs the comparator arm.1 It is the only trial supporting an FDA approval to show superior results against this comparator arm.